DMMS.R – DMMS for Research
TEG’s Diabetes Mellitus Metabolic Simulator for Research (DMMS.R) is a computer application designed for conducting clinical studies in virtual subjects with T1, T2 or Pre-Diabetes. It provides a unique in silico environment for testing diabetes treatment and monitoring interventions, and is ideal for modeling new devices or examining treatment protocols and dosing algorithms. The user can design device models, implement test protocols that rigorously examine the performance of a control algorithm, evaluate the subject response to drug treatments, and assess the performance of sensors and pumps in the in silico environment before clinical testing.
Modeling and simulation of diabetes drugs and treatments early in development can help the user make informed decisions regarding:
- optimal dosing regimens over the full variability of people with the disease
- safety under extreme lifestyle management patterns
- further development to reduce costly failures
Similarly simulation and modeling can aide new drug development. TEG engineers can assist in designing and incorporating proprietary drug or insulin models into a customized version of the DMMS.R.
The DMMS.R is provided with in silico subject populations that represent the variability found in real-world populations. The following cohorts are included:
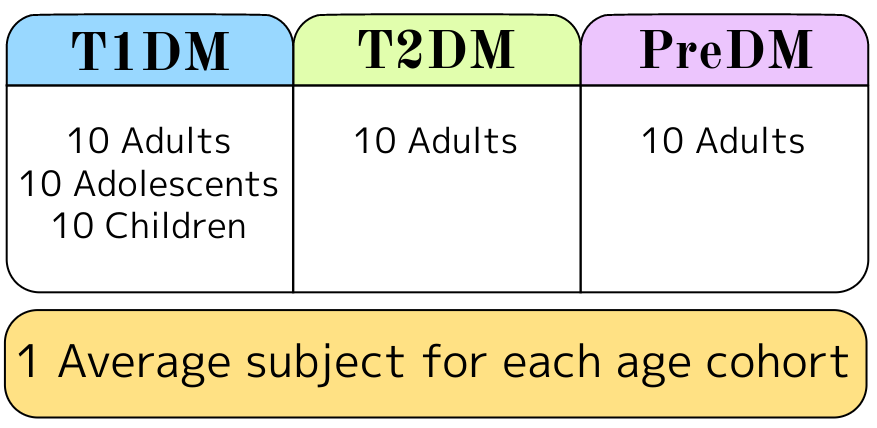
TEG offers further validation of internal in silico study results through simulation services using the 100 subject, FDA-accepted populations. TEG will work with the client to provide custom modeling of devices and drugs, and designing clinical protocols to the client’s specifications.
Simulation results from the FDA-accepted virtual populations can be submitted to the FDA as a pre-clinical study for IDE or 510K approval of new devices, as preliminary data for IND of new drugs or for assessment of bio-equivalence.
System Requirements
- Operating System
- PC: Windows 10 or Windows 11
- CPU
- 64-bit(x64) 1GHz or faster processor
- RAM
- 2GB or more
- Disk Space
- Minimum 50MB of available disk space; better to have some extra space for simulation output
- Screen Resolution
- Minimum 1280 x 800 ; 1680 x 1050 or above recommended
